How to Create a Multi-million Dollar OTT Platform in 2024?
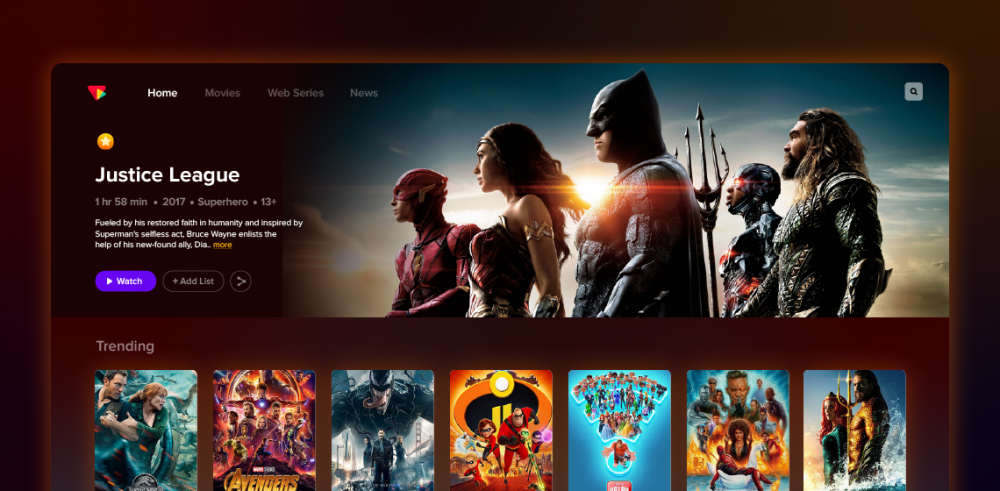
⚡️ Are you looking for the perfect guide on “how to start an OTT platform? Follow these basic steps to create your own platform and monetize your content online. ⚡️
Now is the right time to build your own OTT platform as there are no limitations to the distribution of the OTT content with more content creators turning to OTT production for increasing their audiences up to 30% and their revenue growth.
For all your thoughts on ‘how to create an OTT platform’, here is a 360-degree view on why it is the best idea to get started today.
Table of Contents
What is an OTT platform?
OTT Platform the acceleration of OTT streaming has catapulted video delivery over the internet. With the rapid pace of OTT services bringing substantial changes, there is a considerable impact in the world of media.
“As the OTT revenue is set to approximately hit 167 billion dollars in the year 2025 across the globe, the OTT is touted to be the suitable way to gratify the consumers.”
Based on the type of content delivery and monetization models, the OTT channels are categorized strategically for progressive streaming. OTT services are classified based on their content preferences such as,
- E-Learning
- Gaming
- Sports
- News
- Music
- Entertainment
- Movie
More than 82% of the adult population in the U.S experience video streaming of film, TV, and media content to be more enthralling than cable TVs. Surprisingly, less than 66% of the adult population in the U.S are subscribers of cable TV.
🌟 Read Also: What is OTT? A Definition of OTT & Its Meaning 🌟
Prime Benefits of Building Custom OTT Platform
Content creators can reach their viewers and engage them with video streaming services by creating a unique brand experience. With access to content, your brand gets recognized and familiar amongst the viewers. Modern users have now become more accustomed to the practice of viewing videos anywhere, anytime at the flexibility of devices.
Boosting the Brand Awareness
The OTT videos are efficient in enhancing brand awareness, interest, and perception. Approximately, 70% of the viewers indulge in three hours of viewing videos on a day-to-day basis. A chain of brand awareness, user behavior, etc will bring in more customer loyalty.
More Scalability
The modern technological OTT solution will help businesses launch their OTT platform to extend the measure of the platform and user engagement for increased business profits and ROIs.
Monetization
The prime reason for the OTT service providers to create their own OTT platform is to monetize their video with the availability of multiple business models.
Diminishing the Cost and Time
Building your own online video platform with the help of a white label video streaming provider will be a cost-effective alternative as it cuts down the time for the process and infuses all technological advancements of the solution provider.
Micro-targeting and Data Transparency
Develop your best online video platforms with the fundamental aspects in mind and data encryption with content originality is the major aspect of it. Securely ensure publishing of videos to establish the restriction of geo-localities and work on the persuasion of your audience to watch through a microtargeting strategy.
Convenience
It is mandatory to choose the right niche that seems to be on-demand while planning on how to launch your OTT platform. Sports, eLearning, Spiritual, Fitness, etc are other prominent genres apart from entertainment to explore in the OTT streaming business.
How to Build an OTT Video Platform Successfully?
There is a wide range of OTT services in the market with a unique set of attributes and it is essential to figure out the prime features for comprehensive streaming. You need to consider technological advancements involved in streaming before narrowing down on an OTT platform.
Listed below are the key factors to look out for in the process of building your own OTT video platform.
Step 1. Always pick up a prominent business streaming genre
Pick a suitable genre while creating your own OTT app platform for showcasing the content, On-Demand videos, events, or movies to suit the requirements of the viewers.
Step 2. Target your audiences with Compatibility
Make your audio or video content flexible and compatible for streaming across all platforms and devices with strategic content delivery.
Step 3. Strategize on your resources based on services
Develop your own OTT platform specifically based on the type of content delivery to your viewers. Ensure proper planning of resources to deliver a better viewing experience to your customers.
Step 4. Focus on the improved viewing experience
Deliver original content and focus on delivering a compelling viewing experience. With the availability of multiple OTT providers, concentrate more on streaming valuable content to the masses.
Step 5. Measure your User Behaviour
With a wider and dynamic target audience, it is important to deliver a variety in the content to suit your audience and establish the mindset in the specific demography with analytic tools such as Real-time traffic updates, Current viewership status, User behavior analytics, Revenue Report, etc.
Step 6. Increase ROIs with the right Monetization business models
OTT video services involve live streaming and Video-On-Demand. Pick the monetization model suiting your business needs and it will enable quicker revenue generation by attracting more viewership through advertising. As OTT streaming providers you find ample scope for profitable income in larger divisions.
Step 7. Choose between Websites and Branded OTT Apps
OTT content can be released through an app for viewers or by creating a website. Best OTT platforms should offer tailor-made templates for websites and apps or website builders to create and launch effortlessly.
Make the inclusion of Payment gateways and In-App purchases– Ensure smooth In-App purchases by integrating secure payment gateways into your OTT channel to give your subscribers a hassle-free transfer of money within the application.
Looking To Build Your Own OTT Video Platform?
Start and Grow Your Video Streaming Service With 1000+ Features & 9+ Revenue Models.
Highly Customizable
Life Time Ownership
Own 100% of Your Revenue
Full-Branding Freedom
Progressive Technologies Involved in Creating an OTT Platform
For the generation of increased ROIs from your streaming content, you must infuse the usage of specific tools and stellar technologies to boost the business growth with their progressive functions making them unique and futuristic for content creators and viewers.
I) High performance and Reliable Cloud Hosting
The majority of the OTT service providers use the AWS cloud service for their cloud storage component. Business professionals get benefitted from the cloud-based solutions in building their own OTT platform for increased business revenue and growth.
II) Competent Servers
To handle the larger amount of coexisting connections, make use of the Nginx server. The use of an edge server is undoubtedly the best server in holding the static files and easily streams content in HD quality.
III) Necessary Streaming Protocol
The two prominent streaming protocols put to use in OTT streaming are HTTP live streaming and RTMP. With the use of a Real-time messaging protocol, you can experience low latency interaction and transmission of maximum data.

IV) Superior (CDN) Content Delivery Network
CDN providers are seen as the prime aspects in bringing down the traffic inside the core network and eventually increasing the low latency. The most superior CDN ensures a progressive performance.
V) Robust Security
Security systems are mandatory for OTT streaming success. DRM and AES encryption are the prominent security systems for protecting any content from unauthorized access or copyright issues.
VI) Multi-device access and support
The flexible accessibility across multiple platforms is the right way to ensure the provision of video content at a wide range of devices such as Mobile, Web, Smart TV, Tablet, etc. It is an integral aspect offering the highest scope of business ideas.
VII) IT Infrastructure for seamless distribution
For seamless content distribution, smooth functionality across various platforms is mandatory. The best video streaming will cut down on various other IT resources and make it cost-effective.
Recommended Reading
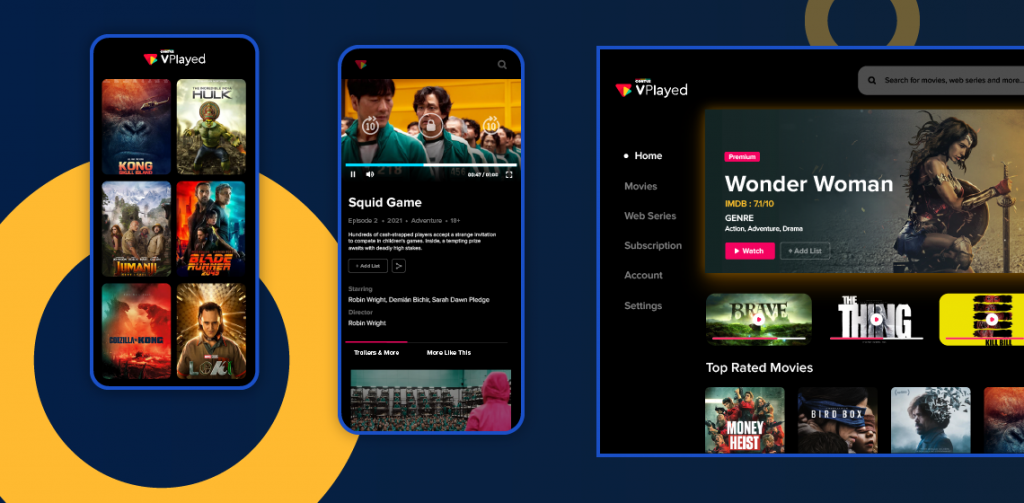
How Does VPlayed help to Create The Branded OTT Platform?
VPlayed shapes your unified streaming platform with the integration of forefront technologies for exploring the comprehensive streaming capabilities. Broadcasters or content creators can empower their audio or video demand with the help of VPlayed with the right monetization model and compatible streaming across devices.
Dive below to explore the outstanding attributes of the VPlayed solution in building your OTT platform.
1. Video Hosting
Experience convenient online video hosting platforms of all your original contents at your liberty with the assistance of VPlayed’s VOD hosting.
2. Video Content Management
With video content management system you can now experience unlimited storage of videos across your channel infused with multiple options like drag and drop, analytics, etc.
3. 100% customizable
Get your best VOD solutions highly customized and end-to-end encrypted services fully integrated to suit your needs.
4. Cloud Transcoding
Integrated with video transcoding will ensure quicker transcoding of content in the process of streaming live content and cloud.
5. Adaptive Streaming
Build video streaming app without any buffering issues at any bandwidth, the adaptive streaming feature will give a complete video streaming experience.
6. Global distribution
Acquire complete access control, and have selective video delivery to strategic locations by monitoring the local content.
7. Content Delivery Network
With CDN networks, manage and distribute the content effectively.
8. Offline download
You can experience affluent offline playback streaming features enabling every user to download any video for offline watch.
9. Monetization Models
With VPlayed, you get the right OTT monetization models for your business with smooth integration of payment gateways and increase your ROIs with TVOD services, AVOD, SVOD, and more.
10. Security
Secure your OTT content with futuristic DRM tools by initiating end-to-end encryption of videos and security protocols to ensure superior safety against digital threats.
11. Channel Partners
Streamline and increase the flow of content by associating with third-party channels.
12. Social Integration
Aggressive social integration will enlarge your audiences and bring in the promotion of videos in multiple social forums.
13. Marketing
Integrate your OTT app with VPlayed’s first-class multi-device compatibility and in-built marketing tools for higher optimization.
14. Analytics
Track data and have an In-depth analysis for decision-making based on the third-party integration with google or Microsoft analytics.
Hire Our Dedicated OTT Developers
Empower your vision of a branded OTT platform with the expertise of our dedicated VPlayed team. With a deep understanding of streaming technologies, we specialize in crafting unified streaming solutions that empower broadcasters and content creators.
Our services encompass the entire spectrum of OTT platform development, from video hosting and content management to adaptive streaming, global distribution, and secure monetization models. You can count on us for 100% customizable solutions, seamless cloud transcoding, and advanced security measures including DRM tools.
Join hands with VPlayed and leverage our expertise to realize your OTT vision. Nevertheless, our skilled developers will build an exceptional streaming experience that engages viewers worldwide, based on your specific requirements.
How Much Does it Cost to Build an OTT platform?
The Cost for creating an OTT platform will be more expensive with the addition and maintenance of more attractive features. You can expect an approximate payment between Fifteen thousand dollars and Two hundred thousand dollars for a branded OTT platform.
Post the development of your OTT TV app plan, you have to focus on hiring the OTT platform developers or bringing in the auto-coding approach. The technology stacks you choose will decide on the price of your broadcasting channel.
Conclusion
As more and more businesses are venturing into OTT streaming, It becomes a herculean task to pick the right solution provider for building your streaming channel. Explore the feature-rich attributes of VPlayed for creating a world best OTT platform with futuristic technological features to increase your revenue generation and double up the business growth.
If you already have an idea about starting an OTT platform, Schedule a free demo and we’ll help you implement it!
Frequently Asked Questions (FAQ)
If you’re thinking about why and how to start your own OTT platform, the important aspect of having access to streaming data and the necessary infrastructure is crucial. This is due to the fact that customers now spend more time than ever before watching video content on their mobile devices. By investing in OTT services, you can take advantage of chances that pay TV can’t provide & directly contact your target audience.
Before you could get into the process of OTT platform development, you should know a bit about the must-haves for any over-the-top based business. Some of the main elements that go into creating your own OTT platform are: complete customization, multi-supportive adaptive video player, access to robust CDN, easy-to-use CMS, branded UI & website, secure payment processing, analytical video metrics, and more.
Like anything else, building an OTT platform has key criteria which play a dominant role in its cost factor. Some of them consist of varied customization, brand objectives along with its target market, platform integration either on cloud/on-premise, and other deliverables that can power up your streaming business such as marketing, analytics, advanced tech-stack like HLS player, centralized CMS & much more.
Accelerate your streaming revenue potential by utilizing the top-most monetization lucrative opportunities offered in the industry. VPlayed lets you have them with a flexible solution that can be tailored to your business niches & audiences. Some of them are Subscription-based (SVOD), Transactional-based (TVOD), or Advertising-based (AVOD). Other than that, you can explore options like Catch Up TV, Coupons & Promotions, Hybrid model, & more.
The OTT entertainment industry has now a plethora of opportunities with the increasing demand for subscription services. Services like Disney+, Amazon Prime, Hulu, Peacock have been drawing enormous revenue upscale, with their creative streaming platform. One who is looking to build video infrastructure needs to know that high-end technologies necessitates investing hugely. Also, platforms need to compulsorily enable seamless content delivery to maintain their ever-growing subscriber base.
As we come to know that OTT video platform are leveling the playing field for independent creators you can actively look into the process of how to create and own one for yourself. Some of the key factors to consider will count with an established audience set to target, regular content addition that can help to sell others existing in the archive transactionally, unique content with a niche following, long-term & making a consistent content publishing calendar.

Such a professional guide & information on creating an OTT platform right from the scratch with necessary details. Good work team.
I am planning to build an OTT platform for my niche based company. Kindly provide me the details for starting from my own service
Is it possible to create an niche based OTT platform with customized features?
Need some suggestion to make a world class ott platform with features like Hulu and multiple revenue models?
Hi, This is David From the USA, We are specialized in creating content in the entertainment sector. So planned to start our own feature-centric ott platform with a seamless delivery option. Is it possible with Vplayed?
Hey I want to know how much does it cost to create an OTT platform with topnotch functionalities like branded platforms
Hey, Can you help us to build a cost-efficient ott platform for our streaming services?
We want to become a successful OTT service provider in our region? Need experts suggestion to start an OTT platform for our niche based service?
Its quite interesting to know about the creating a OTT platform, Need more articles for the various platforms.
Need some details regarding the cost factors for launching a successful OTT platform with drm encrption
Can you please elaborate more about the technical factors used while building a OTT platform and its cost factors to consider in it. Thanks
Is is necessary to make a combined platform and app for our Edu-based services? How to do it with VPlayed solution? Thanks
Hi, I would like to build an OTT platform like Amazon Prime to release independent movies and unique series. Let me know all about starting a OTT platform right from the scratch
I would like to create hybrid OTT platform which will combine transcational and subscription based model. Please I want to talk to someone who can help to start a OTT platform which will be iOS and android friendly.
We are planning to create an OTT platform for our service. Please share the feature list and the quote. We want to launch this in Asia pacific region.
Hello, I have a youtube channel for sports based content. Now I would like to promote it in the OTT platform with multiple revenue generation model. Can you guide us to get start our OTT platform with relevant features.
Hey, One of our client looking for solution to start an OTT platform. Can you share your Tech stack Will you provide the white label with customization features in OTT.
I’ve planned to start an OTT business, Can you please guide to create an OTT platform successfully
Such a useful article! This content has shown exactly how to create an OTT platform. Very informative I must say. Thank you!
It’ll be the perfect time for the content owners to start an OTT platform to stream and monetize it with right model. Invest & earn more on this
I think starting a OTT platform is perfectly suits the corporates and organization.
Great article about creating OTT platform right from the scratch, It lays down very clear guidelines for a successful implementation.
Good Article… You have covered the entire thing in relevant manner, Its one of the perfect choice to start an OTT platform for the business.
Great insights on making a OTT platform successfully, Kudos!
How VPlayed is different from the other platform? What are the unique functionality and benefits on creating a OTT platform with VPlayed? Thanks
We provide media based services to our users at affordable prices for entertainment channels. I am very interested in your service to launch a branded OTT platform. I think your solution is very different from the other providers. It supports Android, iOS & Web Apps.
We are a start-up media company planning to start an OTT platform for our customers to stream entertainment content. We are based in USA, and would like to discuss the different possibilities that we can cooperate together in providing quality content.
I haven’t established a company name yet, but I am trying to see what is possible way to start an OTT platform with the live TV streaming option. Can you please share the option to build out the platform with complete infrastructure.
I am in the process of building my Company branded OTT platform, Would you please guide me through your processes and requirements including your plan pricing structures from stand to enterprise packages. I am working towards launching the platform on this. Thanks Advance
Hello I would like to know if i can open a OTT platform that would be based in USA & Canada? And I’m also cozened about the quality I can deliver through the platform. Thanks!
Looking for complete solution to build White Label OTT platform with AVOD, SVOD and Pay Preview models. It’ll be accessible on multiple devices and smart tv. Which is best way to host our content cloud or on-premises?
We are looking to launch an OTT platform for our US based entertainment business. We would like to see a demo to take things forward. One of our product member approach you regarding this. So kindly schedule a right time for demo session. Thanks
Hai I’m short film maker I want to start my own Ott platform to create the revenue for my short movies pls help me to make this. Need some suggestion from the experts to guide me on right path.
Hi Team, I want to create ott platform for my brand with complete features like Netflix. Moreover I also want to have a back end where the contents can be uploaded by a novice as well.
I’m interested, in how Vplayed may be able to help on how to start ott platform for selling our video content through TVOD and SVOD models.
Greetings, I would like to try out VPlayed for starting OTT platform and also let me know streaming functionality and integrated option with the system. Thanks
For a big client we need to set up and start an OTT platform – many questions option but you seem like a good fit. So kindly arrange a demo session for us.
I am looking to get demo on education purpose, We are one of the leading education industry in USA with 1000+ students enrolled with for their academic purpose. So we decided to start an OTT platform for our institution to support our students. Need you help on how to create ott platform with suitable revenue model for us.
Hi, I’m evaluating options to start an OTT Platform for my company. It must be capable of delivering a smooth streaming experience to our audience and would like to get a demo of the VPlayed platform.
I am looking to make OTT platform that will allow for on demand classes, essentially like a group video so the student can see the teacher, and the teacher can see all the students, is that something we can do? What would the cost look like?
Very useful article. Is it necessary to make a combined platform and app for our video services? How to do it with the VPlayed solution? Thanks
Hello, We are US based art platform and we would like to create OTT platform, an LMS and live concert streaming. Artists should be able to monetize these using advertising and or subscriptions. I was wondering what your company could do for us. Kind regards.
Hi, We are planning to build OTT platform and would like to discuss the service/ product costing and other details. If possible arrange for a demo/ discussion by this afternoon.
Hey, we would love to test your software as we are in the process of building a OTT platform for our services. Already using another one, but not satisfied. So willing to migrate to another third party streaming company. Kindly arrange demo for us. Thanks
Interested in a demo that fully describes this platform to see if its a good fit to create OTT platform for my new streaming business. Also need experts suggestion to make more successful by generation 4x revenue. Thanks Advance
Hello, I am interested in talking with someone to start OTT platform with its prices, storage capabilities, and using your platform(s) for OTT and Livestreaming channels. We have different varieties of channels that we are interested in migrating as well as hosting a permanent channel for some of our other content and live streams. Let me know when a good time to meet for any of you is.
Hello, We would like to explore your solutions to start an OTT platform, as we have projects in the hospitality sector , we are looking forward to have a meeting with you . Thanks and regards,
we need to start an OTT platform customized as per our need for delivering our 200 hrs. of OTT content in USA. Kindly reach out at the earliest with pricing factors and features.
Hi I would like to have a demo of the system to check if it would suit our needs or launching our own OTT platform. Can you please arrange the 1-1 session for us.
We are interested in monetizing our content by starting a OTT platform. Need your suggestion to create, build and monetize our content through streaming platform. Thanks
We are start up in California, We are starting an OTT platform that will broadcast our 6 live TV stations. We will also have series, movies and documentaries that people can watch. we would like to see your platform and discuss options for our requirements. Thank you
Dears, We are searching for the solution to create own OTT platform for our media group. Can you please advise on your pricing Best and demo session. Thanks Advance
Hello, We are one of the top singing academy in the US region. Taking various types of singing classes to our students and recording it for the further batches. Now planned to start an OTT platform for our academy to stream our class recording as per the students convenience with subscription based model. Can you please share regarding this. Thanks
Hey, we would love to test your platform as we are in the process of building a OTT platform for the local tourist market. It showcase the video content of various tourist spots with proper guidance. It will be easy for tourist to know about the place they like to visit. Can you please update the cost factors on this. Thanks Advance.
I am looking to build an OTT platform that will allow for on demand session, essentially like a group video so the students can see the teacher, and the teacher can see all the students, is that something we can do? What would the cost look like?
we need to create an OTT platform customized as per our need for delivering our 200 hrs of OTT content in USA. Kindly reach out at the earliest. Is it possible to showcase the demo session of your platform? It will help’s to make immediate decision for this?
Hello, We would like to start an OTT platform to stream our content across the OTT devices. It should be able to monetize these using advertising and or subscriptions. I was wondering what your company could do for us.
Hi I would like to have a demo of the system to check if it would suit our business needs. We are searching for the perfect platform more than a month, Now satisfied with the features and tech functionality of VPlayed to start our OTT platform. So kindly arrange a Demo session for us asap.
Hello, I am curious about making my own ott platform. How much does it cost on average to set this up? And if possible, may I create sub-channels that are password protected? I manage an event management business and need a way to parse out my individual clients.
Thanks for this comprehensive guide on starting an OTT platform! It’s clear and concise, making it easy for aspiring entrepreneurs like me to understand the process. I appreciate the emphasis on content diversity and the importance of user engagement. Can’t wait to put these tips into action and launch my own OTT platform soon!
Hey, Please let me know the pricing details for starting the OTT platform, & also let me know if we can connect sometime to discuss in detail. We will be having technical questions as well so please pull in technical people in the demo/discussion.
I’ve always been fascinated by the world of video streaming, and your blog has given me the confidence to pursue my dream of starting an OTT platform. The section on monetization strategies was particularly insightful. It’s great to have multiple revenue streams to sustain the platform and provide value to both creators and viewers. Thanks for sharing these valuable insights!
We wanted to make our own ott platform with necessary functionalities like hosting our videos on your video platform and showing that video in our Android and iOS app. There should be an option to add integration and also mid-roll and pre-roll of videos features should be there.
Excellent post! The step-by-step approach in the article is highly beneficial for anyone looking to enter the OTT market. I loved the emphasis on building a strong brand identity and focusing on niche audiences. Your tips on market research and competitive analysis will undoubtedly help new OTT platform owners like me stay ahead of the game. Keep up the great work!
I’m really excited about the idea of launching my own OTT platform, but I was unsure where to start. Your blog has answered all my questions and provided a clear roadmap to follow. The OTT platform development process seems less daunting now, and I appreciate the recommendation to use reliable hosting services to ensure a smooth user experience. Thanks for the guidance!
Hello, I want to build an OTT platform with a subscription model like Netflix. I am looking for a service where I can upload my movies and charge a monthly fee so my customers can watch them anytime they want. Please let me know the right options and price.
We are going to stream our video in high quality and efficient way so we need to create an ott platform with an efficient video player and content delivery for our service.
As a content creator, I’ve been thinking about moving to an OTT platform to reach a broader audience. Your blog has been an eye-opener, as it highlights the various advantages of having to launch an OTT platform. The section on content distribution and marketing strategies has given me valuable insights into promoting my work effectively. Thank you for this valuable resource!
Hi, I am interested in your solution to starting an ott platform with advanced features like Netflix. I would like to know more and watch a quick demo.
Kudos to you for writing such an informative blog post on building an ott platform! I’ve been contemplating starting an OTT platform for my video streaming business, and your article has been a game-changer. Your emphasis on providing a seamless user experience and offering high-quality content is spot on. I’m now more confident in my decision to venture into the OTT space. Thanks for the inspiration!
I had no idea about the technical aspects of starting an OTT platform until I stumbled upon your blog. Your explanation of content delivery networks and adaptive streaming was incredibly helpful. The tips on choosing the right OTT platform provider have saved me a lot of time and effort in research. I can’t wait to implement these suggestions in my project!
I’d like to get pricing to create an ott platform that is just starting up. I currently use Vimeo ott as an experiment and cannot live with their pricing model, Need info about migration too.
I am looking to provide my classes for those who have moved out of town or can not make it into our studio. I am needing pricing to see if this is something I can manage and guidance on how to start ott platform for my service.
Good Day, My team and I are seeking to have our own ott platform similar to Netflix. We’d like to get your assistance and details to build world-class ott service. Thanks
I’ve been thinking about starting an OTT platform to showcase my video content, and your blog has given me the confidence to take the plunge. I especially liked your emphasis on the importance of security measures and copyright protection. It’s reassuring to know that there are ways to safeguard my intellectual property and provide a safe environment for viewers. Thanks for the guidance!
This blog is a treasure trove of information for anyone interested in starting an OTT platform. Your in-depth analysis of the current market trends and potential challenges sets this blog apart from others. I’m grateful for the practical tips on audience engagement and retention. I’m bookmarking this page for future reference as I work towards making my OTT platform a reality!”
Looking for the right service to start an ott platform for my personal training company. It will be a combination of live streaming and video on demand services.
Your blog came at the perfect time for me! I’m currently in the planning phase of launching an OTT platform, and your insights on building a user-friendly interface and incorporating analytics tools were invaluable. It’s great to know the importance of data-driven decision-making and how it can lead to the platform’s success. Thanks for sharing your knowledge!
Just now find your guide on how to build ott platform with profiatbel sources, We are looking for these kind of guide to make a world class OTT platform for our business that is secured, and functional.
Interested in seeing a demo and presentation of your platform. I need proper assistance to create an OTT platform with encrypted DRM features and multi device support.
We need to develop custom a OTT platform with full packages. Kindly share me the cost details for the entire package to start OTT platform with streaming suppor on selected devices.
Hey! We are a start-up and want to launch an OTT platform. We want a subscription-based system with the special feature, that the users can decide themselves which movie they want to give part of their subscription money to.
Hey, Please let me know the pricing details for starting the OTT platform and also let me know if we can connect sometime to discuss in details. We will be having technical questions as well so please pull in technical people in the demo/discussion.
We wanted to create own ott platform with necessary functionalities like host our videos on your video platform and show that video in our Android and iOS app. There should be an option to add integration and also mid roll and pre roll of videos features should be there.
Hello, I want to launch an OTT platform with subscription model like Netflix. I am looking for a service that I can upload my movies and charge a monthly fee so my customers can watch them anytime they want. Please let me know the right options and price.
We are going stream our video with high quality and efficient way so we need to start an ott platform with efficient video player and content delivery for our service.
Hi, I am interested in your solution to make ott platform with advanced features like Netflix. I would like to know more and watch a quick demo.
I’d like to get pricing to build ott platform that is just starting up. I currently use vimeo ott as an experiment and cannot live with their pricing model, Need info about migration too.
I am looking to provide my classes for those who have moved out of town or can not make it into our studio. I am needing pricing to see if this is something I can manage and guidance on how to start ott platform for my service.
Good Day, My team and I are seeking to have our own ott platform similar to Netflix. We’d like to get your assistance and details to build world class ott service. Thanks
Looking for the right service to start ott platform for my personal training company. It will be the combination of live streaming and video on demand services.
Hello, I am curious about creating my own ott platform. How much does it cost on average to set this up? And if possible, may I create sub-channels which are password protected? I manage a event management business, and need a way to parse out my individual clients.